|
Draw the resonance structures for benzene.Ī Each hydrogen atom contributes 1 valence electron, and each carbon atom contributes 4 valence electrons, for a total of (6 × 1) (6 × 4) = 30 valence electrons.Subtract this number from the total number of valence electrons in benzene and then locate the remaining electrons such that each atom in the structure reaches an octet.Create a System of Equations, One Per Element Ba: 1 a 0b 1 c 0d Cl: 2. Sulfur dioxide is a colorless gas with a characteristic choking odor. Sulfate ions in solution, SO 4 2 -, are detected using barium chloride solution. In acidic solution, the equilibria are shifted to form sulfurous acid, resulting in the evolution of SO2 gas. Then calculate the number of valence electrons used in this drawing. Sulfite ion is a weak base, but does undergo some hydrolysis to produce basic solutions. Therefore, more accurately, Li2SO4 is the chemical formula for the ionic compound that is formed when the sulfate ion ( SO42, a polyatomic anion) and the lithium ion ( Li 1, the cation formed when lithium ionizes) bond with one another. Draw a structure for benzene illustrating the bonded atoms. These elements bond with one another as ions, not as neutral atoms.My Website: It’s cable reimagined No DVR space limits. This is a polyatomic anion having a negative charge of -2. This chemistry video explains how to draw the lewis structure of the sulfate ion SO4 2. Given: molecular formula and molecular geometry Sulfate ion (SO42-) is one of the most common ions that people in chemistry need to deal with. Use resonance structures to describe the bonding in benzene. or sulfate ions and its associated co-conformational mechanostereochemical switching. Health effects / safety hazards: The hazards and health effects depend on each sulfate.\)) consists of a regular hexagon of carbon atoms, each of which is also bonded to a hydrogen atom. flexible 2catenane controlled by specific ionic guests. In electricity as the copper sulfate or in water treatment as the barium sulfate. They are largely used in constructions as the Epsom and gypsum salts. 
Uses: Sulfates are present in many natural compounds, for example in rocks, minerals and salts. Thus, the sulfate ion have 6 bonds instead of the typical 4 bonds for smaller atoms. On the other hand, the C-O bond is formed by a sigma bond. This blog post will help you understand if this ion is polar or nonpolar, although a -2 charge might confuse you. Valence electrons are electrons located in the outer shell. On the periodic table: Sulfur, 6 valence electrons Oxygen also has. Elements in this group have 6 valence electrons. Lets do the SO4 2- Lewis structure, for the sulfate ion. It also has a -2 charge because of the additional electrons it accepts to attain this structure. Sulfate Structure Both sulfur and oxygen are found in group VI of the periodic table. Most of them are soluble in water, except barium, calcium, lead, radium, strontium and mercury sulfates.Ĭhemical properties: Sulfate ion is formed due to the sulfur atom has an hypervalency, the S=O bonds are formed by a sigma and π bonds. SO42- is a chemical formula for Sulfate ion it comprises one Sulfur Atom and four oxygen atoms. In general they are solid, mostly powders. Physical properties: Sulfates vary in form according to the salt they are forming. For example, the preparation of metalic sulfates is made from the reaction of metalic salt (in this case barium chlorate) or the pure metal and sulfuric acid:īa(ClO 3) 2 H 2SO 4 → 2 HClO 3 BaSO 4 Preparation: Sulfates can be prepared with a wide variety of processes and depending on the metal to which sulfate is bond. Occurrence: Sulfate ion is largely found in nature as forming different salts and minerals with sodium, calcium, potassium and others. 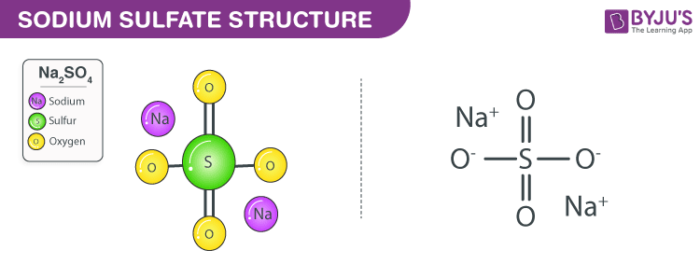
Its chemical structure can be written as below, in the common representations used for organic molecules. The geometry of the anion is tetrahedral. Why Is this because all bonds are equivalent, and. These last oxygen atoms are the responsible for the negative charge (-2) of the anion. My chemical bonding professor says that the O S O bond angles in SO A 4 A 2 are ideal (109.5). This salt is formed by one sulfate center to which 4 atoms of oxygen are attached, 2 of these atoms are forming S=O bonds and the other two are S-O - bonds. Formula and structure: The sulfate ion formula is SO4 2- and the molar mass is 96.06 g mol -1.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
